Services
RSV (Respiratory Syncytial Virus) Vaccination
Arexvy (manufactured by GSK) is the first ‘respiratory syncytial virus’ (RSV) vaccine for adults authorised in the UK, EU and USA.
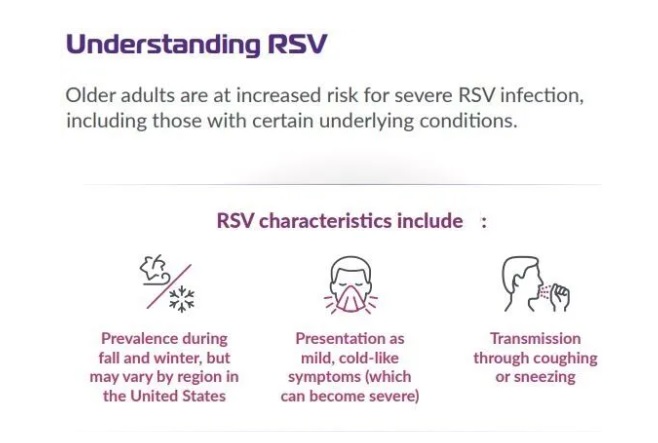
In the UK, RSV infections typically occur in the autumn and winter, and begin to offset in the spring.
There is no treatment for RSV, other than supportive care, for older adults.
There is a substantial burden of RSV infection in older adults, which contributes to the seasonal winter pressures for the NHS.
CDC recommends RSV vaccine for older adults.
Arexvy is given as a single dose injection to protect adults aged 60 years and older against RSV.
Arexvy may be given at the same time as a flu vaccine, in a single visit.
It's advisable to observe at least a 7 day interval with other vaccines.
Trial findings

Safety
- The local administration site adverse reactions reported with Arexvy had a duration of 2 days on average.
- The systemic adverse reactions (fatigue, muscle aches and pain, headache, joint pain and fever) reported with Arexvy had duration ranging between 1 and 2 days.
- Similar rates of serious adverse events (4.2% vs 4.0%), deaths (0.4% vs 0.5%), and F (0.3% vs 0.3%) were reported between participants who received Arexvy (n=12,467) and placebo (n=12,499), respectively.
RSV is a common respiratory virus that usually causes mild cold-like symptoms but can be serious in vulnerable people including babies, older adults, and those with underlying conditions such as diabetes.
This vaccine authorisation from the medicines regulators is based on data from a randomised, placebo controlled trial in 25 000 adults aged over 60 in 17 countries.
The study is ongoing and will be used to evaluate the efficacy of a single vaccine dose over multiple seasons, the need for re-vaccination, and the vaccine’s safety profile. GSK is also carrying out a clinical trial that aims to expand the population who may benefit from RSV vaccine to adults aged 50-59, including those with underlying comorbidities, and results are expected this year.
Andrew Pollard, director of the Oxford Vaccine Group, which is involved in research into RSV, told The BMJ, “RSV is a major contributor to winter pressures in the NHS, usually just prior to the influenza season, with up to 30 000 hospitalisations in infants every year in the UK. The virus also precipitates admissions among frail and elderly adults: the true figure is not certain, but some studies have suggested that the numbers are of a similar magnitude to influenza, at least in some seasons.
“The arrival of new vaccines for adults and interventions to protect babies—maternal vaccination in pregnancy or monoclonal antibodies given at birth—could significantly ease the impact of this virus in the years ahead.”
(from an article in the BMJ: https://www.bmj.com/content/381/bmj.p978)
See the official insert of this vaccine here.
GSK press release: https://tinyurl.com/39j479dh
